Some people think that “stainless steel” is “steel that does not rust.” In their opinion, If it rusts, it must be a fake and shoddy product. This understanding is not quite correct. The “stainless” of stainless steel is not absolutely rust-free, but is relatively rust-free under certain conditions. If the user uses it improperly or it is used in an environment or condition that exceeds its corrosion resistance, it will rust.
It is well known that 304 stainless steel (also known as 18/8 stainless steel) products are food grade, healthy and harmless to human being. 304 stainless steel drinkware and cookware are usually the first choice for consumption. But how do you confirm that the stainless steel you purchased is made of 304 stainless steel?

Click and read more about stainless steel water bottles: 18/8 Stainless Steel Bottle Tumbler Manufacturing Process, Custom Vacuum Hydro Flask Cost,
There are indeed cases of producing and selling inferior stainless steel products on the market. How can we quickly identify these deceptive behaviors when purchasing stainless steel products? Is there any easy way?
Physical Identification means that in the case of an unknown steel grade (steel type), according to the inherent physical and chemical properties of the commodity, simple appliances are used to determine whether it is a stainless steel or a specific type of stainless steel.
It should be pointed out that, Physical Identification cannot distinguish the specific steel grade (steel type), and can only basically distinguish three major categories of chromium stainless steel, chromium nickel stainless steel and chromium manganese nitrogen stainless steel. The identification methods are as follows:
Identification by Color
After pickling, the surface of the stainless steel is bright and white. The chrome-nickel stainless steel is silver-white and jade-colored; the chrome-stainless steel is slightly grayish and slightly glossy; the chrome-manganese-nitrogen stainless steel has a slightly lighter color than the chrome-nickel stainless steel.
The surface color of stainless steel without pickling: chrome-nickel stainless steel is brownish white, chrome stainless steel is brownish black, and chrome-manganese-nitrogen stainless steel is black (the three colors refer to heavier oxidized color).
Identification with a Magnet
The magnet can basically distinguish between two types of stainless steel. Because chrome stainless steel can be attracted by magnets under any condition. Chromium-nickel stainless steel is generally non-magnetic in the annealed state, and some may be magnetic after cold working. However, high-manganese steel with higher manganese content is non-magnetic; the magnetic condition of chrome-nickel-nitrogen stainless steel is more complicated: some are non-magnetic, some are magnetic, some are non-magnetic in the longitudinal plane and magnetic in the transverse plane. Therefore, although the magnet can basically distinguish between chromium stainless steel and chrome-nickel stainless steel, it cannot correctly distinguish some special-purpose steel grades, and it is impossible to distinguish the specific steel grades.
Identification with Copper Sulfate
Remove the oxide layer on the steel, put a drop of water, rub it with copper sulfate, and if it does not change color after rubbing, it is usually stainless steel. If it turns purple, the non-magnetic steel is high manganese steel, and the magnetic steel is generally ordinary steel or low alloy steel.
For special steel grades, we also need to use the following three methods for identification.
Grinding
The identification of the grinding is to grind the stainless steel on the grinder and observe the spark. If the spark is streamlined and has more dense knots, it is a high manganese or manganese nitrogen steel with higher manganese content. If there is no knot, it is chrome steel or chrome-nickel stainless steel.
Annealing Method
For cold-worked chrome-nickel stainless steel, if it is magnetic, we can take a small piece and put it in a fire until it is reddish, let it cool naturally or put it into water to anneal. Generally, after annealing, the magnetic properties will be significantly weakened or completely disappeared. However, some chrome-nickel stainless steels, such as Cr18Ni11Si4AlTi steel and Cr21Ni5Ti steel, have a large proportion of ferrite in their steel, and a considerable part of their internal structure is ferrite. Therefore, it is magnetic even in the state of hot working.
Chemical Qualitative Identification / Stainless Steel Identifying Reagent
The best way to correctly distinguish stainless steel grades is to use stainless steel identifying reagent to identify them. This is also the easiest way.
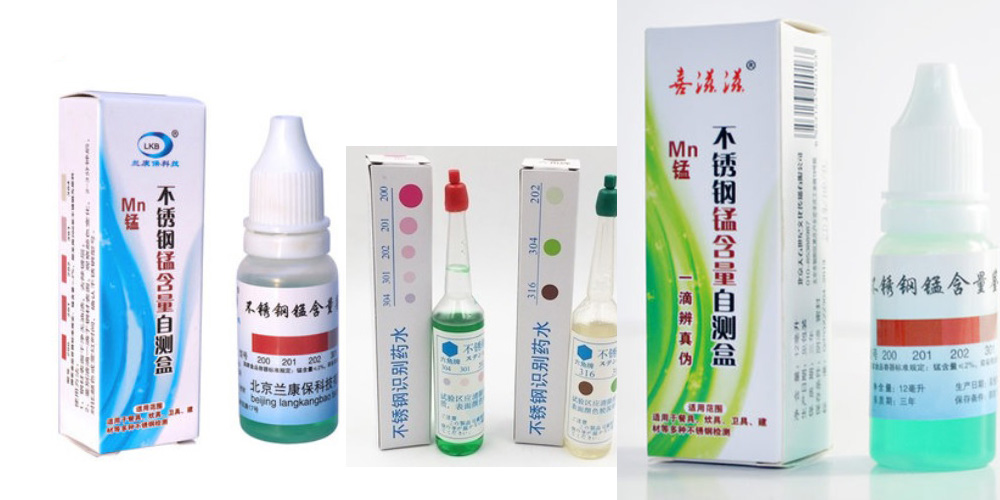
The method of using the stainless steel identifying reagent, is to determine or distinguish by observing the color change characteristics. “Color change” is often associated with specific elements such as nickel (Ni), molybdenum (Mo), and manganese (Mn) in the steel being tested. At present, there are many stainless steel identifying reagents on the market. In Shanghai, Ningbo and other places, some manufacturers specializing in the development of stainless steel identifying / test reagent have been in market for a long time, while some of them have been engaged in chemical technology. Chemical reagent companies have also developed stainless steel identifying solutions for the needs of the stainless steel industry. At present, although the stainless steel identifying reagent has many brands, the products seem to be the same, and can basically be divided into two types. One type does not need a battery and the other one needs to be equipped with a battery.
Test method (no battery reagent): Drop the stainless steel identifying reagent on the surface of the stainless steel, observe the color change of the reagent after two minutes, and then distinguish the stainless steel grade.

The stainless steel identifying reagent that does not require the use of batteries is often not subdivided. The use of such small steel identifying reagents is mainly by measuring the change in color of the droplets after the surface of the stainless steel, and then distinguishing them by the chromatogram. For example, there is a stainless steel identifying reagent called “304” on the market, which is marked with four standard colors corresponding to four stainless steel grades – 201, 202, 301, 304, of which 201 corresponds to deep red, 202 corresponds to red , 301 corresponds to light red, and 304 corresponds to colorless or light yellow. These colors refer to the colors respectively exhibited when the stainless steel identifying reagent is used for the test.
There are many kinds of identifying reagents on the market that need to use batteries, such as “Mo2, low Ni, Ni2, Ni4, Ni6, Ni8, Ni14, Ni20, Ni40, Ni60”, etc. Some products use “N” instead of “Ni” on the label. “. These products can be used alone or in combination to determine the approximate content (percentage) of the corresponding elements of nickel (Ni), molybdenum (Mo), manganese (Mn) in steel, and then to distinguish steel according to relevant standards.
For example, the method of using “Ni8” identifying reagent. When using, firstly apply a proper amount of “Ni8” medicine on a clean steel surface, then connect the positive electrode of the battery to the steel plate, and connect the negative electrode to the “Ni8” medicine bead on the steel surface. Do not touch the steel surface. After a few seconds, stop energizing to observe the color change of the bead on the steel surface. If the color turns red, it indicates that the nickel in the steel is around 8% or above, otherwise the color of the bead will become yellowish or unchanged.
For example, the use of “low Ni” identifying reagent. “Low Ni” identifying reagent is a powerful weapon for the determination of low nickel (Ni) high manganese (Mn) stainless steel. If the bead is purple-red after electrification, the manganese content in the steel is More than 6%, the nickel content is generally below 5.5%. If the bead is colorless or light yellow, generally the stainless steel should be 301, 304, 430 or other materials.

For example, the use of “Mo2” identifying reagent. If the red complex formed by electrooxidation does not fade, it indicates that the molybdenum content in the steel is about 2% or more.
The usage method and color indication of each type of identifying reagent will be described in detail in the instruction manual.
Using the stainless steel identifying reagent, we can easily identify inferior stainless steel. For example, to identify 304 stainless steel, we can use the stainless steel identifying reagent for testing. If it is true 304, we use the above “304 type” identifying reagent or “Ni8” type classification reagent to test, it should appear corresponding test results have appeared corresponding color changes. Otherwise, it is not true 304.
The stainless steel classification reagent on the market is small in size, light in weight, easy to carry, low in cost, and easy to test. It is an effective tool. If you want to test if the stainless steel water bottle you have purchased is made of 304 stainless steel, you can try the method described in this article. As the world’s leading stainless steel water bottle manufacturer, we recommend that you identify product quality and stainless steel grade when purchasing stainless steel water bottles and other stainless steel products.
Order Custom Water Bottles & Bulk Tumblers
Get competitive price and premium custom drinkware from KingStar — Short lead time, door-to-door global shipping, strong Engineering support!
Mr. Samuel Liu is Senior Research&Development Engineer of KingStar Industries Co., Ltd. Please feel feel to send email to info@waterbottle.tech if you want to talk with him on technology and manufacturing.






